-
SONG E-Newsletter December 2017 Edition


As we near the holiday season, it is a good time to reflect on the year that was – and especially to thank everyone who has generously contributed their time, expertise, and effort, which has helped to extend the reach and impact of the SONG Initiative. This edition of the newsletter will provide some recent highlights and updates, and opportunities to be involved!
IMPLEMENTATION
“Patients and clinicians want to know about the effects of interventions on outcomes that matter to them.”
Dr Jonathan Craig, SONG Executive Committee Chair
So far, two core outcome sets – one for haemodialysis (SONG-HD), and one for kidney transplantation (SONG-Tx) – have been established. We are developing strategies and action plans for implementation so that these core outcomes, which are critically important to patients and health professionals, are used in trials to inform decision-making. Of note, researchers can add other outcomes that may be relevant for their specific study.
Implementation workshop
Over 80 patients, family members, clinicians, policymakers, regulators, funders, and industry from around the world attended the SONG Implementation workshop on the 3rd November 2017 in New Orleans. We heard from a fantastic panel, which included the patient, professional society, triallist, regulator, and industry perspective, who collectively provided thought-provoking insights on implementation. The attendees participated in breakout groups to discuss ways to encourage the use of core outcomes in research, particularly in clinical trials. The discussions were diverse and dynamic, with many fantastic ideas and suggestions to move forward. A full report of the results from the workshop will be published in 2018. 
Multi-stakeholder break out discussion groups at the New Orleans workshop – brainstorming opportunities for implementation of the core outcomes.
“The SONG Initiative workshops are a great way to learn from patients, care partners, physicians and other experts while working together to improve the science of kidney disease research. I participated in two SONG workshops in New Orleans at Kidney Week and really loved how the healthcare professionals ensured that I understood everything that was said during the discussions. I never felt talked down to, I felt like a partner.” David White, Patient Partner
Endorsement
To date, the SONG initiative has been endorsed by 16 organisations worldwide, and most recently by the International Society of Nephrology, The Transplantation Society, Kidney Disease Improving Global Outcomes (KDIGO), the Renal Association UK, and the Cardiovascular and Renal Clinical Trialists Network (INI CRCT).
SONG-HD HAEMODIALYSIS
It is full steam ahead as we work towards developing, piloting and validating core outcome measures for each of the SONG-HD core outcomes (fatigue, vascular access, cardiovascular disease, and mortality).
Fatigue 
SONG researcher Angela Ju, with Executive Committee Member and Patient Tess Harris, presenting the SONG-HD fatigue workshop at the American Society of Nephrology Kidney Week.
In our systematic review of patient-reported outcome measures for fatigue used in research in haemodialysis, we found that 43 different measures have been used across 123 studies, but none appear to have been validated in patients on haemodialysis. Based on the results of the review, survey, and consensus workshop (Chicago, 2016), we have now developed and piloted a core outcome measure for fatigue (questionnaire with three questions) among patients on haemodialysis to make sure that patients understand the measure and agree that it covers the most important dimensions of fatigue (i.e. impact of fatigue on life participation) identified in the survey. The international validation study will begin early next year. Vascular Access  SONG researcher Dr Andrea Viecelli explains her poster on vascular access measures currently used in trials to a very engaged audience at Kidney Week in New Orleans.
SONG researcher Dr Andrea Viecelli explains her poster on vascular access measures currently used in trials to a very engaged audience at Kidney Week in New Orleans.From 168 recent trials included in our systematic review of vascular access outcomes reported in hemodialysis trials, 1426 access related outcome measures were used for 23 different vascular access outcomes, which again points to the need for standardised reporting of this outcome. Based on the international survey and consensus workshop (Chicago, 2016), vascular access function was identified as the core outcome, which may be defined as “need for intervention/ procedure to maintain the use of the vascular access for dialysis”. Once we have finalised the above definition for vascular access function and its metric (i.e. “time to event”, “number of events over 1000 access days”), we will evaluate the feasibility and reliability of the proposed measure in an international pilot study. CVD  The amazing patients who joined us in New Orleans all the way from Houston for the SONG-CVD consensus workshop with Executive Committee Member Dr Wolfgang Winkelmayer.
The amazing patients who joined us in New Orleans all the way from Houston for the SONG-CVD consensus workshop with Executive Committee Member Dr Wolfgang Winkelmayer.Based on an international online survey with approximately 500 patients, caregivers and health professionals, heart attack and sudden cardiac death were identified to be the most critically important cardiovascular outcomes to be reported in all trials in hemodialysis. The results were discussed at the recent SONG-HD Cardiovascular Consensus Workshop held on the 2nd November, 2017 in New Orleans, organised by SONG researcher Dr Emma O’Lone. Over 60 patients, caregivers and health professionals attended and there was very interesting discussion and debate, particularly around the use of heart attack (myocardial infarction) and/or sudden cardiac death as the core outcome measure for cardiovascular disease. A workshop report will be available in 2018.
SONG-Tx Kidney Transplantation
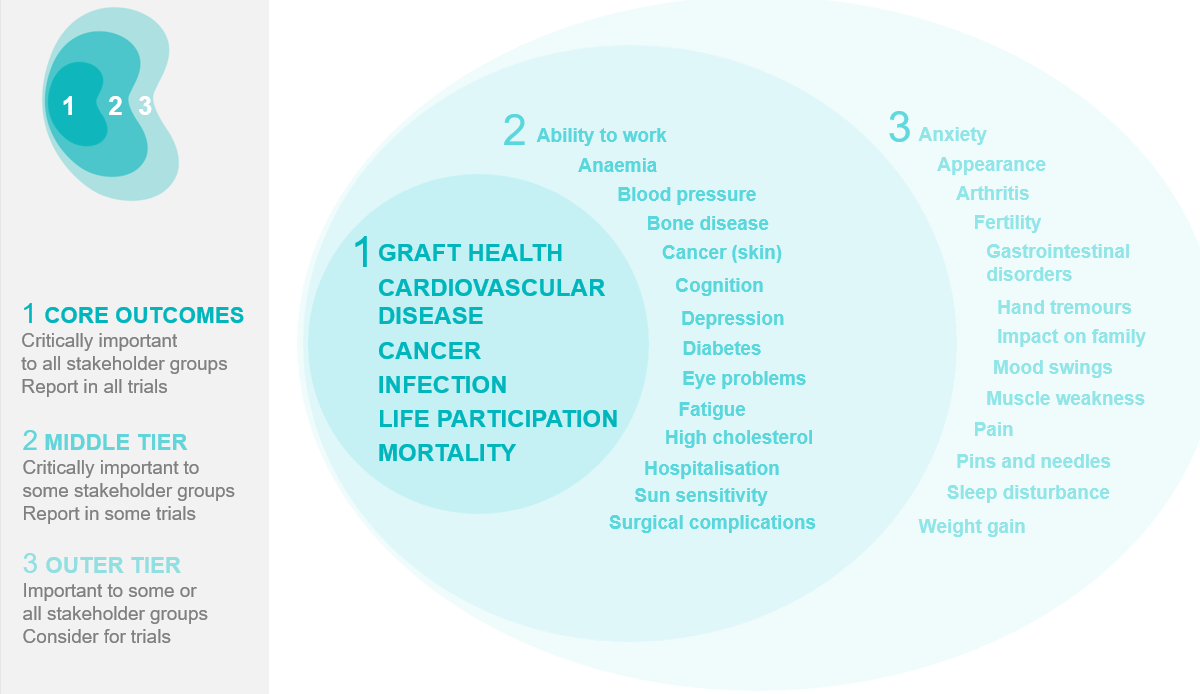 We are currently working on a suite of systematic reviews to identify current measures used for life participation, graft loss, cancer, and cardiovascular disease in trials in kidney transplantation, to inform the selection or development of core outcome measures for these core outcomes.
We are currently working on a suite of systematic reviews to identify current measures used for life participation, graft loss, cancer, and cardiovascular disease in trials in kidney transplantation, to inform the selection or development of core outcome measures for these core outcomes.Graft loss
Based on preliminary results of the systematic review and consensus workshops (Chicago, 2016), the proposed definition for the core outcome measure for graft loss is: “chronic dialysis for more than 90 days or repeat kidney transplant.” The workshop report has been submitted for publication. Once the measure has been finalised, we will pilot and evaluate the measure.
SONG-Kids Children and Adolescents
 SONG researchers Laura James, Camilla Hanson and Talia Gutman with a wonderful group of parents and kids who participated in the Nominal Groups in Melbourne, Australia.
SONG researchers Laura James, Camilla Hanson and Talia Gutman with a wonderful group of parents and kids who participated in the Nominal Groups in Melbourne, Australia.We completed the SONG-Kids focus groups in Australia, the United States and Canada, and in total, 34 children and 62 parents were involved in identifying important outcomes for children with CKD and their families. Based on the preliminary results: for children, the top five outcomes were: sport, kidney function, fatigue, infection and survival; and for parents the top five outcomes were: kidney function, weight gain, survival, infection, and anxiety/stress. The outcomes currently reported in trials in children with chronic kidney disease, and the outcomes that were identified by children and their parents to be important, will be included in the international Delphi survey to be launched next year. If you are interested in participating in the SONG-Kids Delphi survey, please register here.
SONG-PD Peritoneal Dialysis
We conducted the final round of focus groups in Hong Kong in September, after running groups in the United States and Australia. In total, we ran 14 groups involving 126 patients and caregivers. The SONG-PD Delphi Survey was recently launched in October this year. In total, 207 patients/caregivers and 666 health professionals from 69 countries completed the first round of the online Delphi survey.
So far, the top five outcomes that were determined to be critically important to all stakeholders were PD related infection, death, membrane functioning, PD failure, and cardiovascular disease. Thank you to everyone who completed round 1!
 SONG researcher Karine Manera with patients contributing to the SONG-PD focus groups in Hong Kong.
SONG researcher Karine Manera with patients contributing to the SONG-PD focus groups in Hong Kong.
SONG-PKD Polycystic Kidney Disease
 Patients from the SONG-PKD focus groups conducted in Korea earlier this year. Fantastic to have such diverse collaborators for this project!
Patients from the SONG-PKD focus groups conducted in Korea earlier this year. Fantastic to have such diverse collaborators for this project!We are pleased to announce the protocol for SONG-PKD has been published in the journal Trials. This year, we completed 16 focus groups with nominal group technique in three languages with patients and caregivers across Australia, South Korea and France. We are currently analysing the results, which we plan to publish next year.
Patient Involvement
Patient involvement is absolutely critical to the SONG initiative. We are currently interviewing patients and carers that have been involved in research in chronic kidney disease to find out about their experiences and to understand how they would like to be involved in identifying and implementing core outcomes. We also conducted a survey following the focus groups for SONG-Kids, SONG-PKD and SONG-PD to find out from patients and carers how they felt about participating and how we can improve the participant experience. The results from these studies will be published next year and will be used to improve and further enhance patient involvement.
 SONG Patient Partner David White with SONG researcher Talia Gutman at Kidney Week in New Orleans – a star contributor sharing his inspirational passion for patient advocacy at all the SONG events! We are so grateful to the patients and family members who share their voices in our workshops, focus groups and interviews.
SONG Patient Partner David White with SONG researcher Talia Gutman at Kidney Week in New Orleans – a star contributor sharing his inspirational passion for patient advocacy at all the SONG events! We are so grateful to the patients and family members who share their voices in our workshops, focus groups and interviews.We really appreciate your involvement in this community effort to drive culture change, to help improve the relevance, certainty, and value of research for better care and outcomes in people living with kidney disease, and we look forward to continuing to work and collaborate with you in 2018. Our warmest wishes for a wonderful Christmas and New Year, from all of us at SONG!




