SONG-Tx
Why develop core outcomes in kidney transplantation?
Kidney transplantation improves survival, health, and quality of life outcomes in many patients with end-stage kidney disease compared with dialysis. Major advances in treatment have dramatically improved short-term graft survival and acute rejection. However, there are still a number of challenges to the success of kidney transplantation. Kidney transplant recipients need to take medications that suppress their immune system, which can place them at a higher risk of of cancer, cardiovascular disease, diabetes, and infection.
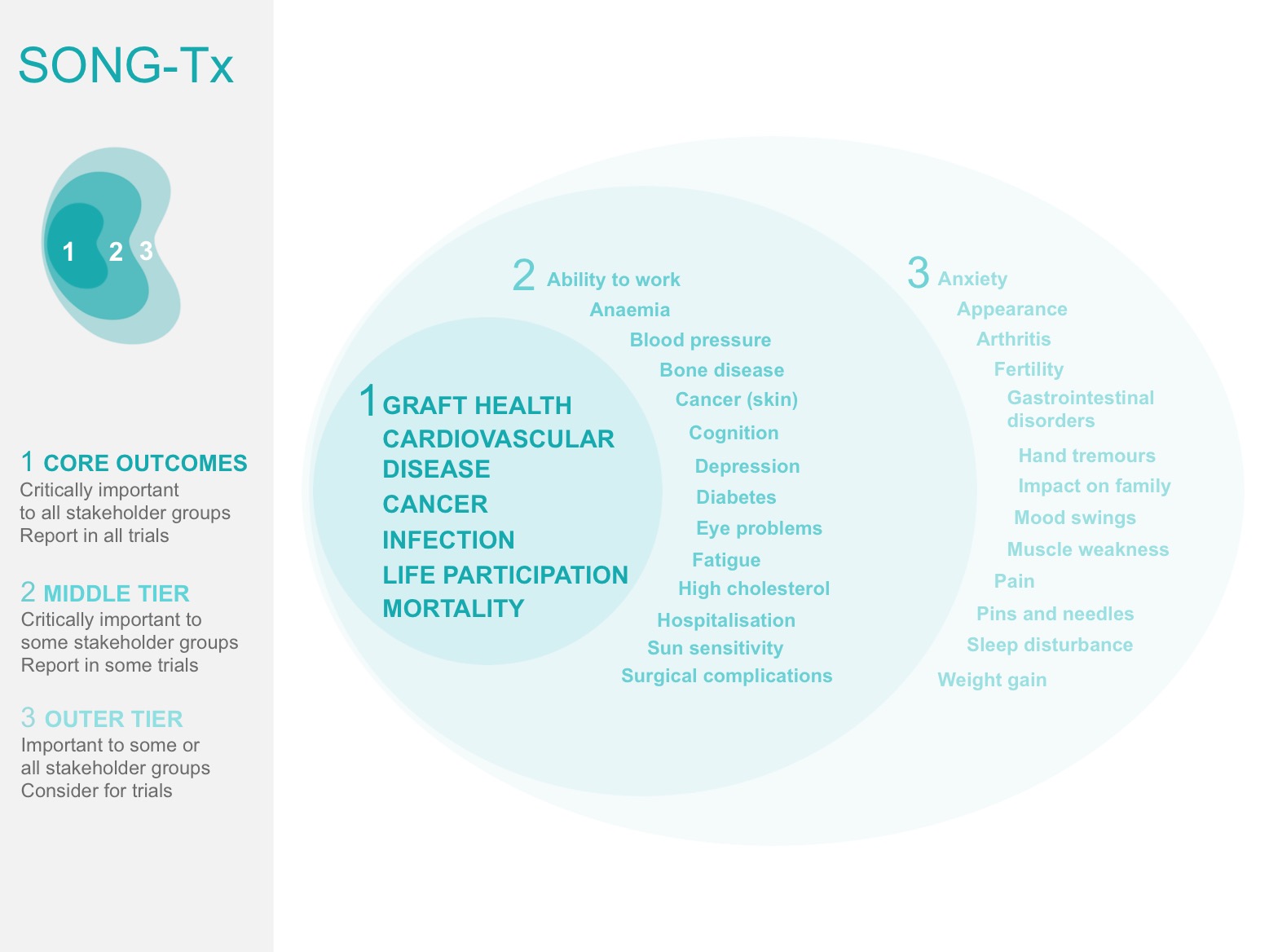
Many clinical trials have been conducted in kidney transplantation but a wide variety of different outcomes are reported and in different ways, and it is unclear whether they are important to people who have received a kidney transplant. There has been no agreement about what are the most important outcomes to measure and report in studies for kidney transplant recipients. The Standardised Outcomes in Nephrology – Transplantation (SONG-Tx) study aims to develop a core outcome set for trials in kidney transplantation that is based on the shared priorities of patients, family members, and health professionals .
SONG-Tx Steering Group
Jeremy Chapman (Chair) | Westmead Hospital, The University of Sydney, Australia
Anthony Warrens | Queen Mary University of London, United Kingdom
David Rosenbloom | Health Services Advisory Group Inc ESRD Network 18, United States
Germaine Wong | Westmead Hospital, The University of Sydney, Australia
John Gill | University of British Columbia, St. Paul’s Hospital
Klemens Budde | Charité – Universitätsmedizin, Germany
Lionel Rostaing | Toulouse University Hospital, France
Lorna Marson | The University of Edinburgh, United Kingdom
Michelle Josephson | The University of Chicago, United States
Peter Reese | University of Pennsylvania, United States
Tim Pruett | University of Minnesota, United States
Identifying core outcomes domains
SONG-Tx involves 3-phases:
- a systematic review to identify outcomes that have been reported in trials in kidney transplantation
- a three-round Delphi survey with stakeholder groups to distill and generate a prioritised list of core outcomes
- consensus workshops to establish a core outcome set for kidney transplantation trials.
Project updates
SONG-Tx Delphi survey
The SONG-Tx Delphi Survey has closed. The Delphi panel included 1018 members (461 patients/family members, and 557 health professionals).
SONG-Tx Consensus Workshops
June 2016 | Boston
The SONG-Tx Boston Consensus workshop was held Monday 13th June 2016 during the American Transplant Congress in Boston. Many thanks to the 68 kidney transplant recipients, family members, and health professionals who engaged in very interesting and thought-provoking discussion on establishing core outcomes for trials in kidney transplantation. (Click photo for larger image)

Attendees: Ajay Israni, Alan Leichtman, Allan Massie, Allison Tong, Allyson Hart, Angelique Ralph, Beatrice Oakley, Benedicte Sautenet, Bert Kasiske, Camilla Hanson, Caren Rose, Chris Watson, Christine Murphy, Christophe Legendre, Dana Basken, David Rosenbloom, David Shakespeare, Devin Peipert, Fritz Diekmann, Gabriel Danovitch, Germaine Wong, Gerry Chipman, Greg Knoll, Hallvard Holdaas, Heidi Basken, Ina Jochmans, James C Varnum, Jamie Wells, Jayme Locke, Jennifer Trofe-Clark, Jenny Shen, Jeremy Chapman, Jessica Ryan, John Gill, John Kanellis, John Mazzullo, John Scandling, Joseph Kacoyannakis, Kjersti Lonning, Klemens Budde, Klemens Meyer, Krista Lentine, Linda Rosenbloom, Ling-Xin Chen, Lorelei Basken, Lorna Marson, Marc Cavaillé-Coll, Matthias Buchler, Michael Germain, Michael Murphy, Nicole Evangelidis, Peter Friend, Peter Reese, Phil Clayton, Phil O’Connell, Rainer Oberbauer, Randall Morris, Renata Albrecht, Robert Bulger, Robert Steiner, Rosemary Kacoyannakis, Roslyn Mannon, Sabina De Geest, Sheila Jowsey-Gregoire, Siah Kim, Sobhana Thangaraju, Stephen Fader, Steve Alexander
Contributors: The full list of attendees and collaborators are listed in the SONG-Tx Boston Workshop Program.
August 2016 | Hong Kong
A second workshop was held Saturday 20th August 2016 during The Congress of The Transplantation Society to discuss the findings and potential core outcome set for trials in kidney transplantation. Building on the key points from Boston workshop, the discussions were largely focussed on graft-related outcomes, quality of life and patient-reported outcomes, and strategies for implementation.






Attendees: Beatriz Dominguez-Gil, Benedicte Sautenet, Benita Padilla, Brian Chu Yuen Tse, Camilla Hanson, Chi Yan Yuen, Choi Fong Hau, Curie Ahn, Deneb Cheung, Dirk Kuypers, Fabian Halleck, Frank Dor, Germaine Wong, Greg Knoll, Hai An Ha Phan, Janet Hui, Jeremy Chapman, Jif Wong, Joen Hui, Jonathan Craig, John Gill, Hatem Amer, Helen Pilmore, Jayme Locke, Jongwon Ha, Kai Ming Chow, Klemens Budde, Kirsten Howard, Lalitha Raghuram, Lin Ping, Lionel Rostaing, Marina Ng, Madeleine Didsbury, Maggie Ma, Martin Howell, Mirjam Tielen, Nga Lun Mok, Nick Larkins, Paul Harden, Penny Allen, Peter Stock, Peter Nickerson, Richard Allen, Romina Danguilan, Ron Shapiro, Samuel Fung, Shigeru Satoh, Stephen McDonald, Tahir Aziz, Teck Chuan Voo, Terence Kee, Vasant Sumethkul, Vathsala Anantharaman, Vivekanand Jha, Allison Tong
Contributors: The full list of attendees and collaborators are listed in the SONG-Tx Hong Kong Workshop Program
A draft workshop report will be circulated to all attendees and contributors.






